Table of Contents
What will you learn in this article?
Mucormycosis and COVID both are today angels of death partnering with each other. Mucormycosis has been an unexpected disaster and is now an epidemic in India. Also know as BLACK FUNGUS, everything seems to be a mystery about this fungal menace.
COVID-19 pandemic itself has been a massacre. Millions have died. And there seems to be no end. Medical science is still learning how to tackle this virus. Treatment is ever evolving. Each day it seems we are better prepared as we keep learning. But we are nowhere close to the complete solution to this challenge.
Around 10% of infected patients land up in hospital.[1] Once you reach that stage where you need hospitalization, it means you are strolling near the thin red line beyond which the darkness of death looms. Survival becomes the key concern now. Most end up coughing out several lakhs of rupees trying to afford for extremely costly medicines such as Remdesivir, Tocilizumab etc. Liters and liters of oxygen and high dose steroids keep running through the veins of these patients in the hope of surviving from the clutches of death.
Many of these hospitalized patients do survive. They come out of hospital with a sense of victory over death. Although battered and crippled both financially and physically, the dawn of fresh new second life looks promising.
But, for some of these lucky survivors, luck stops favoring soon. Dark clouds of hopelessness once again start looming. It starts with numbness around cheeks. Then comes pain around that region and eyes. The pain slowly starts getting unbearable. Fever resumes. It is not easy to diagnose. But the diagnosis of MUCORMYCOSIS is nearly like a death sentence. By the time diagnosis happens, it’s generally too late. Patient has to be hospitalized. Surgery is mandatory. Many lose their eyes. Very few medicines work. The ones that work are rarely available due to shortage in supplies. Even with the best medicines, survival is like toss of a coin- nearly 50% die.
I had already covered this topic briefly in an earlier article– but that was quite brief. In this article we will explore this new epidemic of mucormycosis that is killing COVID survivors. We will try to uncover the real reasons behind this sudden emergence of mucormycosis and why India is home to this menace. We will also try to understand how this disease may be treated in situations where you don’t have access to the current gold standard drug called Amphotericin B. Finally, we will attempt to explore various ways and means of preventing mucormycosis.
Introduction to Mucormycosis

Mucormycosis is a dangerous fungal infection. It is caused by a group of molds called mucoromycetes, which is a saprophytic fungi of the order Mucorales.[2] Rhizopus arrhizus is the most common mold causing mucormycosis in India and globally.[3]
It is usually a rare infection. But, if it happens, it is potentially a fatal infection.[4] This fungus feeds on iron in the blood, eventually destroying the blood vessels. As the blood supply gets cut off, the tissues which depend on those blood vessels die off. Slowly but surely the area of invasion keeps growing and as the fungus keeps destroying the blood vessels more and more tissues keep getting destroyed.
Link between Mucormycosis and India
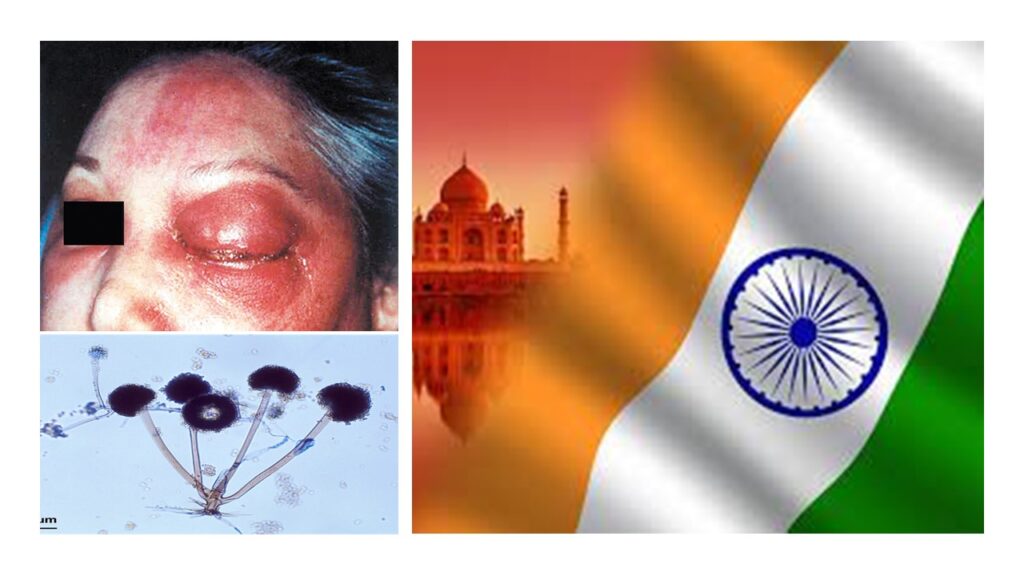
Mucormycosis is not a new disease. It was initially described in 1885 by Paltauf, and was known by various names including zygomycosis and phycomycosis. [5] We, in general, never really heard of this because it’s actually pretty rare.
We have to remember that Mucormycosis is not unique only to India. It is seen across the world. But since we are the world’s largest population in the world, its statistically obvious that India would contribute to a very high number of cases. Globally the incidence rate of mucormycosis is about 0.005 to 1.7 per million population.[6] However, more than 70% of the cases are reported from India. India actually reports highest burden of mucormycosis in the world with an estimated prevalence of 140 cases per million population.[7]
Mucormycosis and COVID- Situation in India
The second wave of COVID-19 has affected India widely and gravely. The number of daily reported cases was a little more than 0·4 million in the first week of May, 2021. Since then, the number of new reported cases has reduced. However, during the third week of May, 2021, India still contributed nearly half of the total new cases detected globally and nearly 34% of the deaths globally.[8]
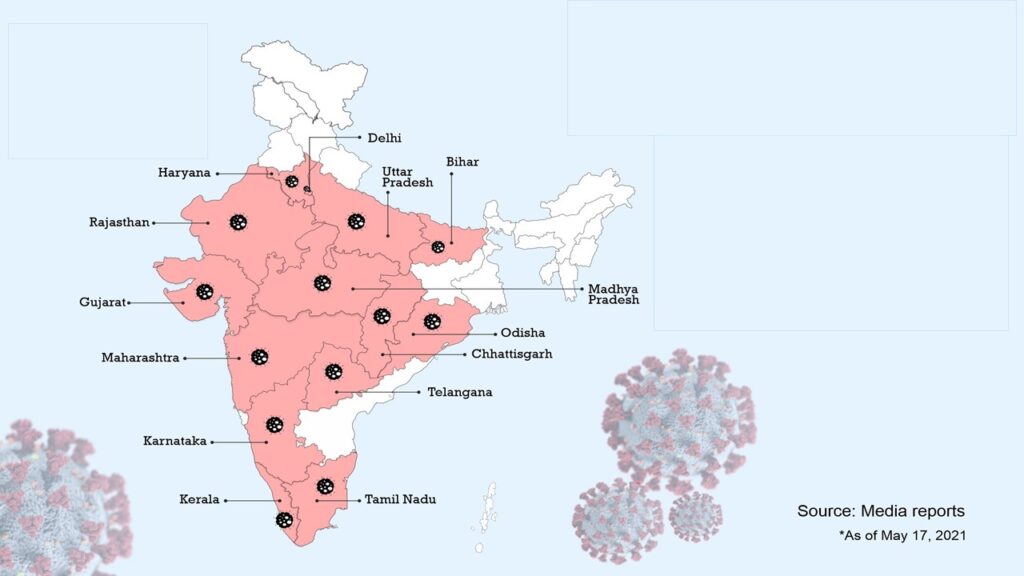
As India struggles to combat the existing situation with COVID-19, mucormycosis is slowly emerging as another dangerous challenge of epidemic proportion in India. Often referred to as the “Black Fungus”, the incidence of mucormycosis has risen more rapidly during the second wave compared with the first wave of COVID-19 in India. As of 28 May 2021, there were nearly 15,000 reported in India. The state of Gujarat contributed to the highest number of these cases. There were at least 3726 cases of mucormycosis in patients with active and recovered COVID-19 in Gujarat alone. The state of Maharashtra closely followed Gujarat in terms of reported cases of mucormycosis. Other states such as Rajasthan, Andhra Pradesh, Karnataka, Haryana, Madhya Pradesh, Uttarakhand, and Delhi have also reported a steady rise in the number of mucormycosis cases and deaths related to it. Many of these states already having declared it as an epidemic and a notifiable disease to the national health authorities.[9]
Link between Mucormycosis and COVID
The most common causes attributed to the rise of mucormycosis in COVID-19 patients are uncontrolled diabetes, the excessive use of corticosteroids for immunosuppression, and long-term stays in the intensive care unit. [7]
However, the link between Mucormycosis and COVID-19 is not that simple to delineate. People have been taking corticosteroids earlier as well- in large doses for long period of time. Uncontrolled diabetes is not new in India- India leads the world in terms of diabetes burden. It’s only in this second wave of COVID-19 that we are witnessing this massive surge of Mucormycosis.
Let’s try to analyze the facts that we know:
- COVID-19 led immune suppression: COVID-19 infection itself makes us susceptible to opportunistic infections like mucormycosis. In majority of the cases, there is significant loss of defense cells called lymphocytes (medically this loss of lymphocytes is known as lymphopenia). This lymphopenia is more pronounced in patients developing severe COVID-19. [10] [2]
- The common door- GRP 78: The fungi causing mucormycosis make use of a receptor called GRP 78 present in our blood vessels for launching its attack. And GRP 78 has been postulated as one of the receptors responsible for entry of COVID-19 virus. [7]
- Role of iron: Fungi feed on iron. They proliferate on the iron available in blood. In COVID-19 the amount of available iron in blood rises. More the severity, more is the available iron. This “available” iron is also reflected by the fact that severity correlates with serum ferritin levels.[11] Similarly, diabetes also leads to increase in iron availability to fungus. [12]
- Diabetes and mucormycosis: Presence of diabetes adds to the risk of mucormycosis in COVID-19 patients very significantly. Diabetes is a common finding in more than 90% of mucormycosis cases in COVID-19. [13] India is home to the second largest pool of diabetics (prevalence ~8.9%) worldwide, with up to 42% of them being undiagnosed.[14] COVID-19 itself causes a “diabetogenic state” in patients- more the severity of the disease, greater is the risk of development of diabetes. [7] Increase in blood sugar increases the level of acidic ions in blood. This is known as acidosis. Acidosis impairs functioning of immune system. Acidosis also causes an increase in iron available in blood, which itself can easily promote growth of mucormycosis.[15]
- Corticosteroids and mucormycosis: Many experts claim that it’s the abuse of corticosteroids for treatment of COVID-19 that has led to emergence of fungal infections such as mucormycosis. But facts say something else. Across the world millions of patients have been taking corticosteroids daily for decades for various conditions such as autoimmune diseases. Forget corticosteroids, thousands of patients across the world have been taking drugs like Tofacitinib for years [16]. Tofacitinib is much more potent immune suppressant than corticosteroids. Of course there are side effects when you take such medications for years, but they are all manageable[17] [18][19]. And of these side- effects, Mucormycosis never emerged amongst the most concerning ones.
- Use of oxygen and mucormycosis: We just have hypotheses here and nothing scientifically proven. But there are few pointers to ponder upon. We have seen much higher incidence of mucormycosis in India during the Wave 2. Iron is an essential component for mucormycosis. To combat acute shortage of oxygen, India might be the only country which has diverted iron industries during Wave 2 to produce and supply oxygen. There iron cylinders come from various sources and lay stacked up in damp unhygienic conditions which are actually ideal for any fungus to proliferate. And the spores will continue to exist, theoretically, even if these cylinders are then filled with oxygen. Is this the reason behind epidemic? Possible, but can’t say for sure. Most cases have been seen in patients who had been admitted to hospital and taken oxygen. So, is the source of mucormycosis spores these oxygen cylinders? Possible, but again can’t say for sure. Oxygen from the cylinder goes through a humidifier before reaching the patient. The water used in this humidifier, if not clean, might theoretically contain fungi and can enter the patient. Again, nothing is proven.
My personal opinion is that fungi and their spores are in the air. If you are susceptible, you don’t need any special source like oxygen cylinders to infect you with fungi. The reason why it’s so commonly seen in hospitalized COVID-19 patients is the very fact that you will be hospitalized if you generally have severe COVID-19.
If you read the above points again and again, I hope you will see the pattern that is now obvious to me. Diabetes is linked with severity of COVID-19 infection. More the blood sugar, more is the likelihood of severe COVID-19. Availability of iron in blood is again directly correlated with severity of COVID-19. High dose of corticosteroids for long duration is seen in hospitalized patients with severe COVID-19. Oxygen cylinders are utilized only in patients with progressive COVID-19 disease.
The combination of hyperglycemia, low pH, high iron, decreased activity of defense cells due to COVID-19 and enhanced expression of GRP-78 receptors on the blood vessels makes it way easier for the mucormycosis fungi to enter and proliferate.[20]
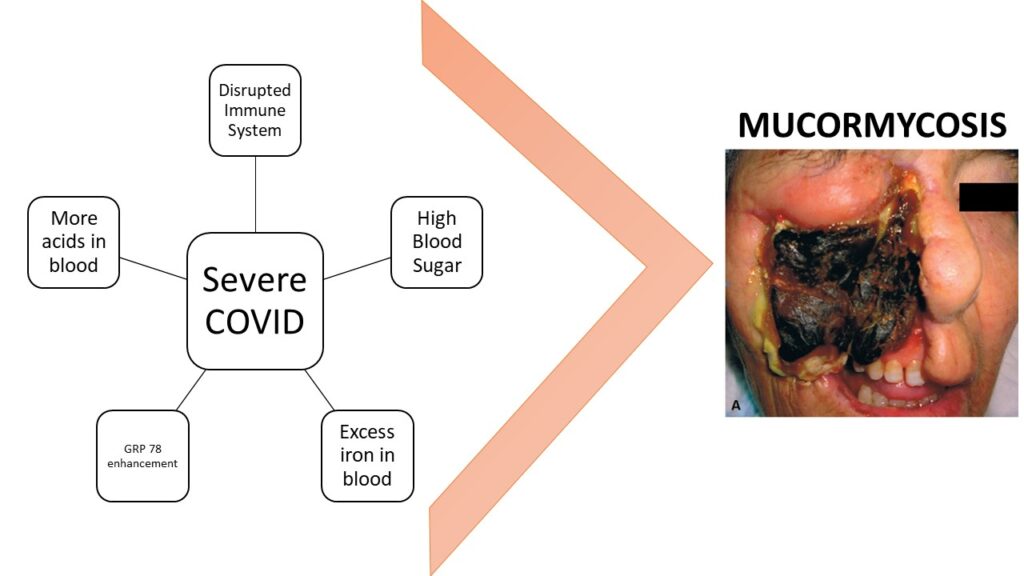
It’s the severity of COVID-19, rather than anything else, that is the main factor amplifying the risk of mucormycosis. Rest everything is just noise.
Early Diagnosis is the key to treat Mucormycosis
Mucormycosis infection is scary- scarier than COVID-19 itself. Across the world, case fatality rate with mucormycosis is close to 50% [5]. Issue is mucormycosis is very difficult to diagnose. By the time it gets diagnosed, its generally too late. Early diagnosis and treatment are essential. A delay of even 6 days is associated with a doubling of 30-day mortality from 35% to 66%.[6]
Its extremely critical to remember following points to aid early diagnosis:
- High level of suspicion: In COVID-19, be vigilant about risk of developing of mucormycosis if one has one has severe disease requiring oxygen or hospitalization. Similarly, if one has diabetes mellitus one must be vigilant. Patients suffering from cancer and undergoing chemotherapy also have heightened risk of developing mucormycosis. Also vigilant should be those who have been on immunosuppressive medications for a long period of time.[21]
- Catching early symptoms: In many patients its usually starts as a combination of one sided headache especially behind eye region, facial swelling/pain, numbness, fever, deranged sense of smell, and nasal congestion. It then progresses into necrotic eschars with black purulent discharge from nasal cavity. There can be patches of black discoloration over skin of nose or persistent stuffiness of one side of nose. In later stages, vision gets impacting starting with blurriness, diplopia progressing into complete loss of vision. Soon the fungus will invade brain and death is not far away. This form of mucormycosis which starts by invading nose, sinuses and facial region is known as rhinocerebral mucormycosis. [21] [22] Some patients start by complaining of pleuritic pain. Pleuritic pain refers to sudden and intense sharp, stabbing, or burning pain in the chest when inhaling and exhaling. Such pain is exacerbated by deep breathing, coughing, sneezing, or laughing. These patients also can complain of fever, breathlessness, and cough. These symptoms indicate another form of mucormycosis known as pulmonary mucormycosis. In many cases rhinocerebral and pulmonary forms co-exist. [21] [22]
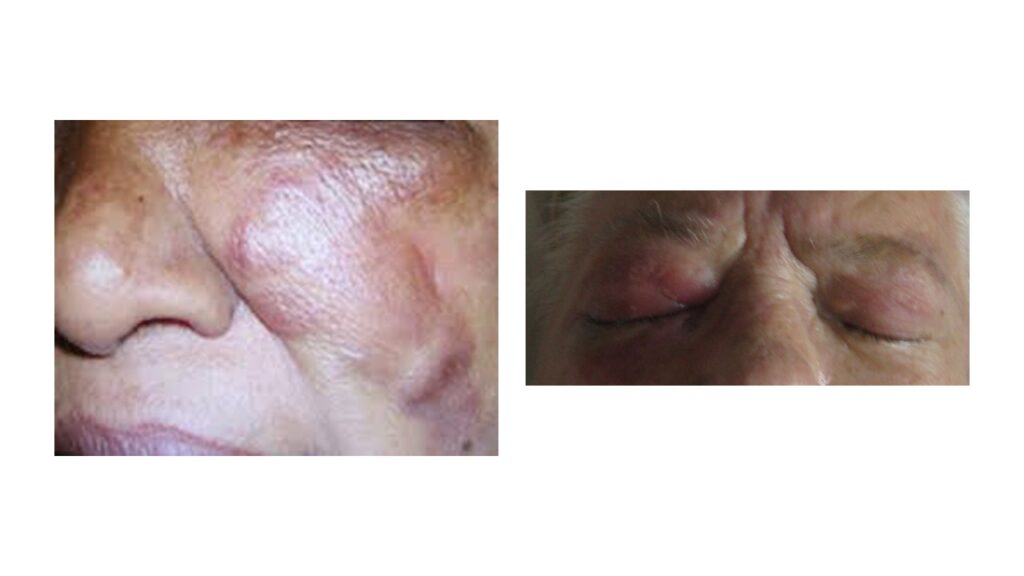
So, if one has complaints resembling sinusitis after COVID-19 infection, immediately suspect mucormycosis. Similarly, if one has fever, breathlessness and chest pain after recovering from COVID-19 infection, one must rule out mucormycosis.
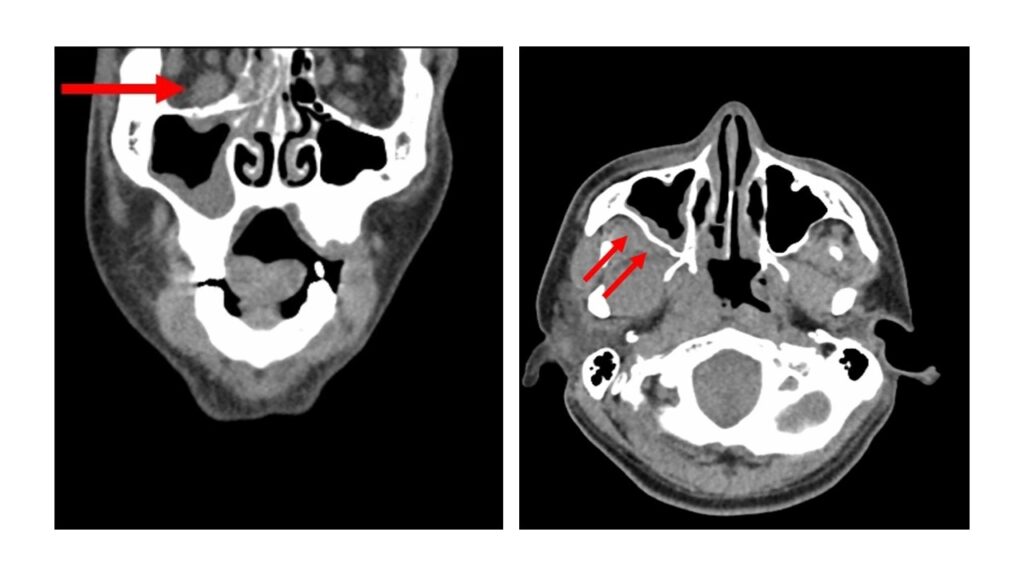
- Radiology: Earlier recognition of mucormycosis can be aided by CT scanning and is way better than normal X-rays. In patients with very high index of suspicion, CT scans can actually reveal development of mucormycosis even before classical symptoms emerge.
(This following paragraph in italics is for those who can understand radiological terminologies. Others may skip.)
Nonenhanced high-resolution CT scanning is the imaging modality of choice for diagnosing mucormycosis. Classical findings include pleural effusion, nodules, consolidation, and ground-glass opacities. As the disease progresses, consolidation may become multilobar. The reverse halo sign (ie, a nodule with central ground-glass opacity and a ring of peripheral consolidation) is a strong indicator of pulmonary mucormycosis. The halo sign (ie, a nodule surrounded by ground-glass opacity) represents a lung infarct surrounded by alveolar hemorrhage. This sign is associated with invasive mold infections. However halo sign can be present in bacterial or viral infections and noninfectious lung diseases such as wegener’s granulomatosis, sarcoidosis, malignancy etc. Other findings such as the air crescent sign and hypodense sign have been described- but these are less specific and may occur in later stages.
- Biopsy: Biopsy of the involved tissue is required for establishing a diagnosis of mucormycosis. A rapid histologic assessment of a frozen tissue section is critical so that one can promptly institute surgical and medical management for the infection. Microscopic examination after using appropriate stains reveals the typical broad (typically 6- to 25-µm diameter), irregular, ribbon like fungal fibres which are sparsely septate and which branch irregularly at 45-90 degrees angle.[23] Culture of biopsy sample is another way of confirming mucormycosis. It usually takes around 2-3 days for the fungus to grow in the culture media. But remember- it’s actually quite difficult to demonstrate fungus via culture. The fungus quite fragile and gets easily damaged. [24][23]
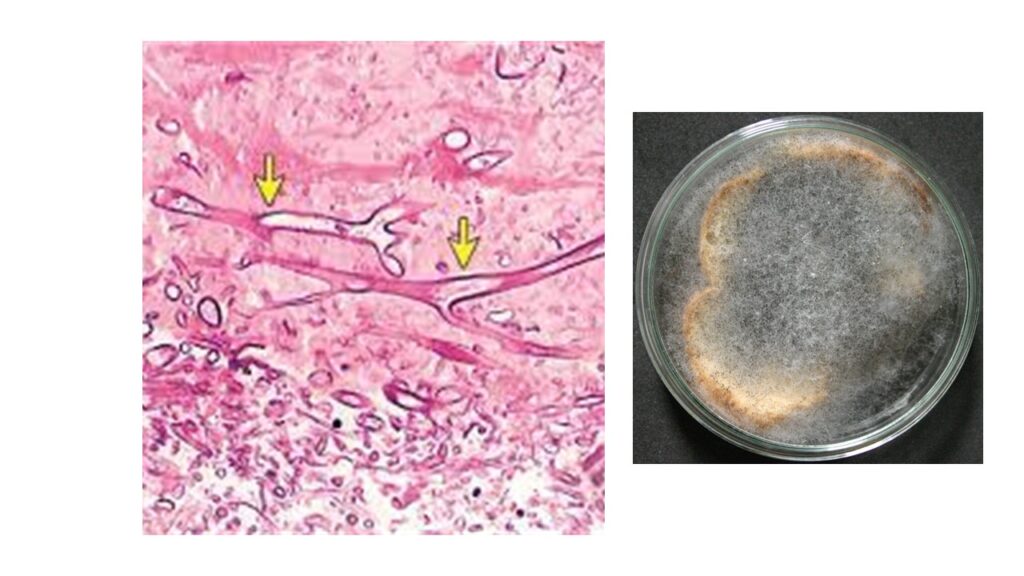
Histological evaluation via microscopy is more sensitive than culture. Cultures may be negative even if histopathology confirms mucormycosis. [24]
Treatment of Mucormycosis
We saw that early treatment is critical and any delay incrementally increases the mortality rate. However, one must note that even if its diagnosed early and treatment is promptly initiated, prognosis still remains poor despite aggressive combined surgical and medical therapy.[6]
The recommended treatment line is surgical debridement, and prompt administration of intravenous antifungals – usually Amphotericin B.
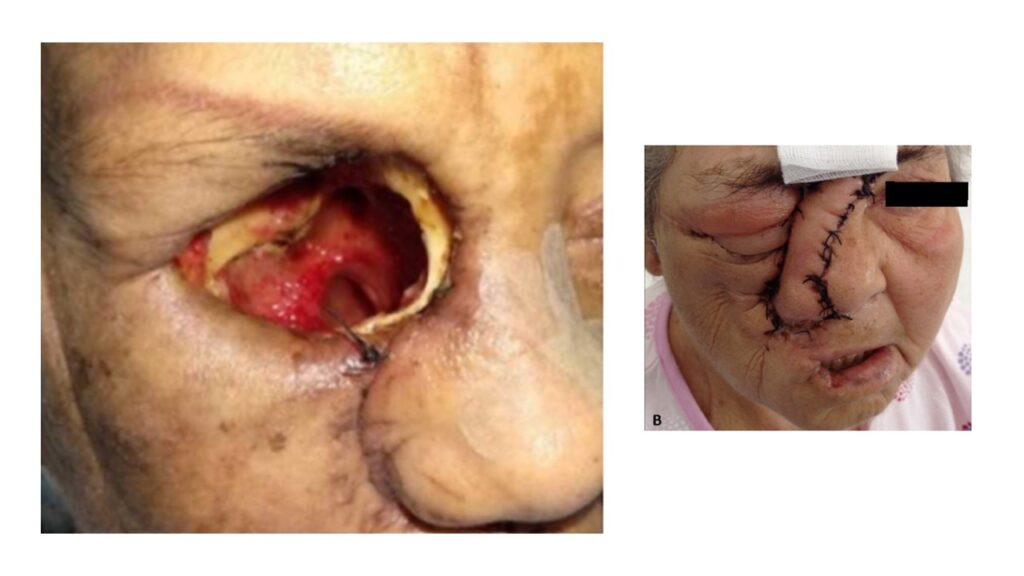
It is very important to limit further spread of infection. This cannot be achieved just by medicines alone. Surgical debridement of infected tissue should be done urgently and effectively. [25] This is quite scary for the patients. Most cases are quite advanced, and many a time involve surgical removal of eyes and maybe a portion of jaw leading to permanent facial disfigurement. Unfortunately, there are no good alternatives.
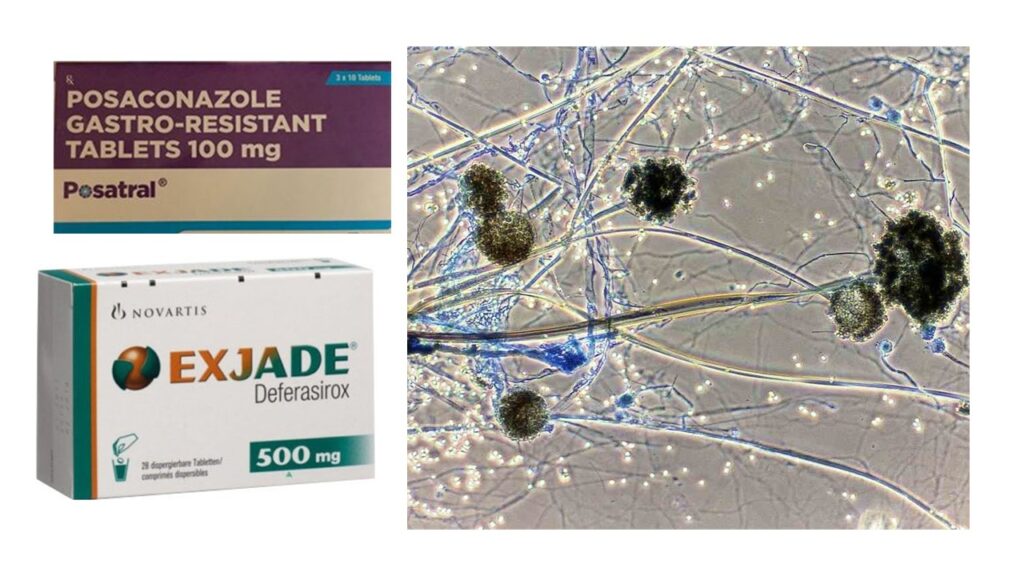
Mucormycosis is resistant to most known anti- fungal drugs. There are only 3 antifungals that have shown to work- Amphotericin B (Amb) and its lipid formulations, Posaconazole and Isavuconazole.
The first-line recommended antifungal agent against mucormycosis is Amphotericin B (Amb). Although, Amb is the is highly toxic to kidneys it is the most active drug available against mucormycosis. Isavuconazole and Posaconazole are the other alternatives. However, none of them are as effective as Amphotericin B. And given the fact that even Amphotericin B fails to be life- saving in many cases, its unlikely any doctor will take chances by trying Isavuconazole or Posoconazole. Infact, mucormycosis cases have been reported in patients undergoing Posaconazole prophylaxis despite satisfactory serum concentrations.
Combating mucormycosis, in case Amphotericin B is not available.
Amphotericin B has been widely recognized as the only drug that works against mucormycosis, along with surgery. Due to this, there is today an acute shortage of Amphotericin B across India.
What if a patient is suffering from mucormycosis and Amphotericin B is not available?
Fighting against any pathogen is like defending your territory in a war. The army of pathogen is trying to invade your body and destroy it, and your aim is to prevent the pathogen from doing so. One way is to use weapons that can destroy the pathogen. But what if the enemy is very powerful and you don’t have weapons that can effectively kill its army?
Scorched earth policy is a military strategy which involves destroying anything that might be useful to the enemy which is advancing through an area. All of the assets that can be used by the enemy are made useless or unavailable, such as food sources, transportation, communications, industrial resources, and even the people in the area. In 1941, when Germany attacked Russia, Russia was unprepared to counter that invasion. They did try to put up some resistance, but Germans were too powerful and couldn’t be stopped. It was then Stalin adopted “scorched earth policy”. The philosophy was- a hungry enemy with no resources can’t invade much. Essentially, the Soviet troops burned their own crops, destroyed bridges, and evacuated factories in the face of the German advance. Entire steel and ammunitions plants were dismantled and were shipped away from the German reach. Even the rail system around the path of German advance were destroyed. The scorched earth policy created severe problems for the German war machine which was trying to keep her three million soldiers supplied with the necessary food and ammunition.[26]

So, is it possible to starve mucormycosis and we cannot kill it?
And the answer is- maybe YES. We earlier discussed quite in detail that the mucoromycetes (the fungi causing mucormycosis) feeds on iron present in blood. If this is made unavailable, mucormycosis cannot spread. The central role of iron as primary requirement for mucormycosis infection leads to the theoretical possibility of utilizing effective iron chelators as adjunctive antifungal therapy. When tested in labs, it has been conclusively shown that iron chelators like Deferasirox did not allow the mucormycetes to take up iron, ultimately killing the fungus.[27] When administered to diabetic mice and mice with compromised immunity infected with mucormycosis, Deferasirox significantly improved survival and decreased tissue fungal burden, with an efficacy similar to that of Amphotericin B.
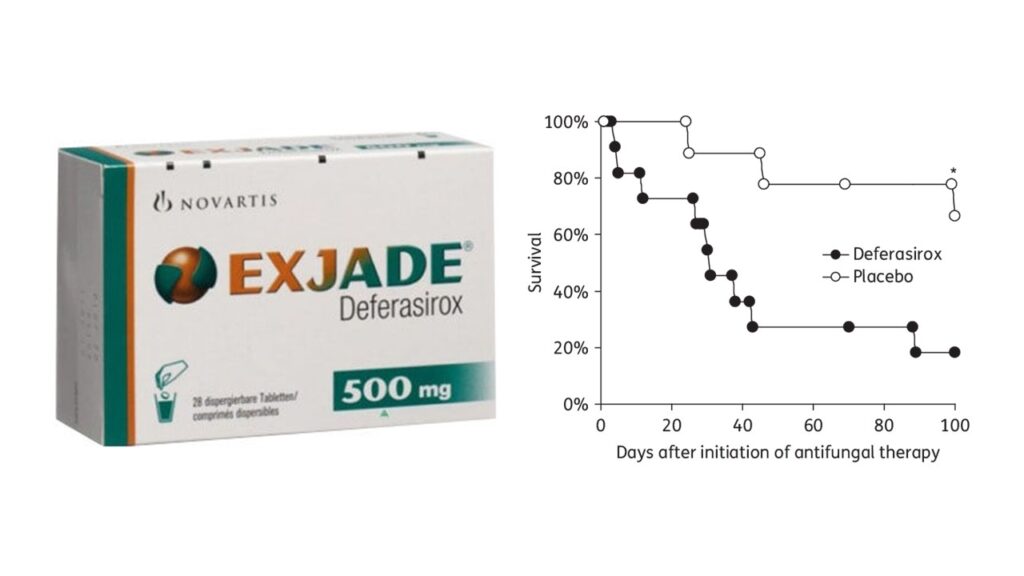
A 2011 study showed how compassionate use of Deferasirox helped cure 7 patients with mucormyosis in whom complete debridement was not possible.[28] Patients seriously infected with mucormycosis and not responding to Amphotericin B might respond and get cured if Deferasirox is added. [29]
We already saw that Posaconazole is active against Mucormycosis, although not as effective as Amphotericin B. Posaconazole has enhanced in vitro activity against the Mucorales (the fungi that cause mucormycosis. Posaconazole has a reported 90% minimum inhibitory concentrations (MIC90) of 1 to ≥ 4 μg/mL.[30] Available clinical data do suggest that posaconazole is a reasonable option for patients with mucormycosis who are refractory to or intolerant of Amphotericin B.[31]
Deferasirox is oral. Its easily available in India. Posaconazole is also oral. And, the above data makes me believe that, if there is no access to Amphotericin B, Deferasirox may work miracle when combined with Posaconazole.
I have to make a humble declaration here- I have no idea if Deferasirox + Posoconazole works in mucormycosis. I have no experience. I was lucky that no one got this scary mucormycosis infection. The combination I am suggesting maybe tried in extreme situations when you have no access to any hospital. And again, you would need a doctor’s supervision.
Prevention of Mucormycosis post COVID
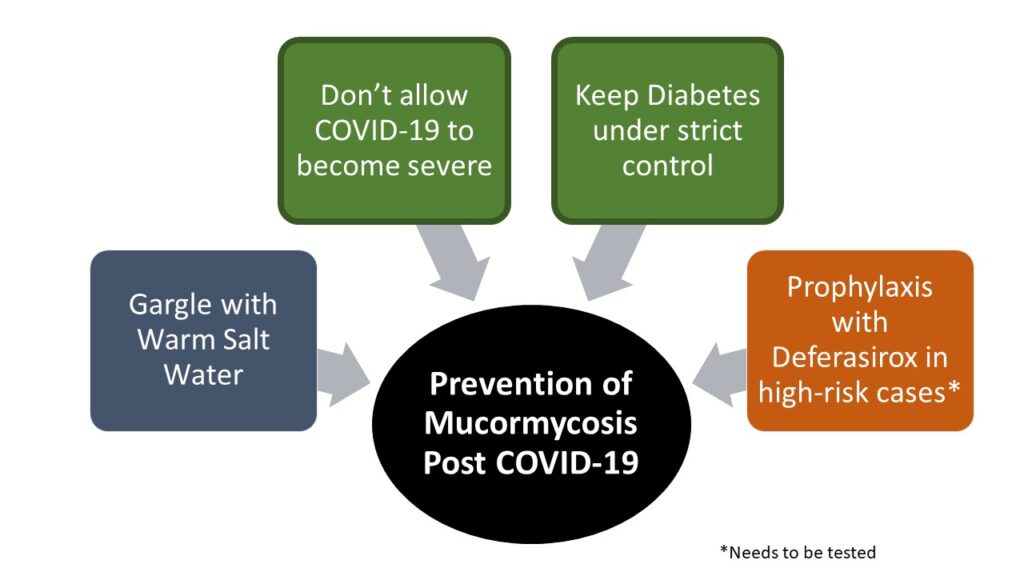
Based on my understanding of pathogenesis of mucormycosis, I would recommend following to prevent mucormycosis.
- Do not allow COVID-19 to become severe: Treating doctor must identify early signs that indicate the disease might become severe and take steps to prevent the same. This might mean early use of corticosteroids for a short period after Day 6 in presence of early “red flags”.
- Keep diabetes under control: Each patient with COVID-19 must be checked frequently for blood sugar levels. These levels must be kept under check aggressively
- Other unproven ways:
- Gargles with salt water: I recommend gargles with lukewarm water mixed with salt, thrice daily to all patients with COVID-19, from Day 1. The chloride ions from salt is a power anti-bacterial and anti- fungal and might protect against secondary infections.
- Prophylaxis:
- Prophylaxis with antifungals should be strictly avoided. Antifungals are toxic and can cause serious side effects. Also, there is a risk of developing resistance, which can then lead to fungal infections that won’t respond to antifungals.
- However, in extremely high-risk cases, it’s possible that prophylaxis with Deferasirox might help. 200 mg per day for 30 days might turn out to be an effective strategy to prevent mucormycosis in high risk patients. This strategy just ensures the fugus doesn’t get iron to feed upon and grow. And 200 mg for 30 days should be safe enough. Adequate safety has been established with much higher dosage of Deferasirox taken daily for around a year. [32] This hypothesis regarding role of Deferasirox in preventing mucormycosis needs to be urgently tested.
Mucormycosis post COVID- My Concluding Remarks
Mucormycosis is really scary. There are lot of theories around why the incidence has gone up during COVID-19. Most of these are just theories with no evidence. What we do know for sure is COVID-19 disrupts our immune system, and more severe the disease higher is the chance of mucormycosis. Similarly, higher the level of blood sugar, higher is the chance of this fungal infection. So, if you are infected with COVID-19 make sure your blood sugar is under control and you ensure the disease doesn’t become severe. In worst case scenario, if infection does occur and Amphotericin B is not available, maybe Posoconazole + Deferasirox might work. Lastly, I really feel Deferasirox should be studied to check if it can used to prevent Mucormycosis in high risk cases. It seems to a very safe and effective way to ensure protection against Mucormycosis.
References
[25] J. L. Hernández and C. J. Buckley, Mucormycosis. StatPearls Publishing, 2021.
[26] “Scorched Earth.” https://spartacus-educational.com/RUSearth.htm (accessed Jun. 04, 2021).





Interesting facts ….always looks forward to leatn from uour articles..SOCRATES
Thanks Vinod!
I am glad that you liked it!
Attractive section of content. I just stumbled upon your web site and in accession capital to assert that I acquire actually enjoyed account your blog posts. Anyway I’ll be subscribing to your feeds and even I achievement you access consistently fast.
Thank you for you compliments!
Wspaniały blog wiele przydatnych informacji zawartych w poszczegolnych postach, Dzieki Ci za to serdeczne, zapraszam także do siebie.
Your article helped me a lot, thanks for the information. I also like your blog theme, can you tell me how you did it?
I agree with your point of view, your article has given me a lot of help and benefited me a lot. Thanks. Hope you continue to write such excellent articles.
Your article helped me a lot, is there any more related content? Thanks!
Really informative post.Really thank you! Awesome.
I love foregathering useful information , this post has got me even more info! .